Membrane potentials as being a property of all cell membranes
Resting membrane potentials as a consequence of asymmetric distributions of Na and K (Note: detail of the specific ionic and voltage changes is NOT required)
Action potentials as being a property of excitable tissues
A potential difference across the membrane (outside the cell vs inside the cell) is known as the membrane potential [Em].
This varies between cell types.
In excitable tissues, resting Em is usually between –60 and –90 mV (Like pacemaker cells).
The muscle cells it is around -90mV (milli volts)
Asymmetric ion distribution
Cell contains molecules that are positive and negative.
Negatively charged molecules like proteins cannot cross the membrane (the fixed negatives).
These negative charges attract positive ions like Sodium and Potassium. But membranes are more permeable to K+ than Na+ leading to potassium accumulation inside the cell.
This accumulated K+ concentration gradient drives K+ back out of the cell.
Ultimately, the cell remains negatively charged, causing a potential difference across the membrane.
Equilibrium state of electrical forces Vs concentration forces is Gibbs–Donnan equilibrium.
K+ Equilibrium potential, (EK) can be calculated from the Nernst equation.
Intracellular [ K+ ] = 120mmol/L and extracellular [ K+ ] = 4 mmol/L, EK = ∼–90 mV.
Intracellular [ Na+ ] 10mmol/L and extracellular [ Na+ ] 140 mmol/L, the ENa = +70 mV.
The permeability of different cells to K+ (PK) to Na+ (PNa) ratio ranges between 25:1 and 100:1 in nerve, skeletal and cardiac muscle cells. (Cells have 25 to 100 times more permeability to potassium in comparison to Sodium)
The Em at rest (resting membrane potential) is close to EK (–60 to –85 mV) and the electrochemical gradient for K+ is small.
If PNa is suddenly increased, the sodium will rapidly enter inside along electrochemical gradient and Em would shift towards ENa . (During an action potential, the Na+ channels open to increase the permeability ( PNa ) ~10-fold and the membrane becomes positive – depolarizes.
The depolarization is primarily due to increase in permeability to ions which leads to changes in concentration.
Appreciation of the ionic basis of the action potential
Depolarization – Importance of the threshold potential in initiating propagation
– Propagation based upon the all-or-nothing principleRepolarization – Ionic basis of the absolute and relative refractory periods
Curriculum
Depolarization
The initiation of Action potential in nerve and skeletal muscle starts when the neurotransmitter activates Sodium channels. This type of channel is known as ligand-gated Na+ channels.
The post synaptic Sodium channel is Ligand gated in muscle cells. Not voltage gated.
Frequently tested.
The channel opening increases permeability to Sodium (PNa) and causes influx of Sodium ions, resulting in EM moving towards ENa (i.e. become positive).
If the stimulus is strong enough to make the cell cross the threshold potential (∼−55 mV), Voltage-gated Na+ channels will get activated, and Sodium enters in large numbers making the cell voltage positive.
Em reaches towards ENa (∼+65 mV).
This sharp positive ‘spike’ is DEPOLARIZATION – lasts about 1 ms in nerve and skeletal muscle.
Depolarization lasts about 300ms in cardiac cells, which is why cardiac muscle doesn’t show tetany
Clinical pearl

Repolarization
Once Em becomes positive, the voltage-gated Na+ channels get inactivated (PNa drops, no more sodium entry) and voltage-gated K+ channels get (delayed rectifier) activated leaking potassium out reducing the voltage.
The Em returns towards EK reaching baseline negative value as before depolarization. This is called as repolarization.
As repolarization continues, the delayed rectifier K+ channels remain open for some more time, leaking more potassium than required, causing a transient hyperpolarization (excess negativity).
Once initiated, the action potential can travel only in one direction with same amplitude (it is all-or-nothing).
Refractory period
After depolarization, the Na+ channels remain inactive until the cell is largely repolarized and, during the early period, no matter how strong the depolarization is, they cannot be opened. This is known as the absolute refractory period.
Impossible to generate another action potential in this period.
Following the absolute refractory period, the transient hyperpolarization makes the cell difficult to depolarize again. It’s difficult but not impossible. A larger stimulus is needed to generate an action potential. This is the relative refractory period.
Pacemaker potential

In pacemaker cells, funny current or slow Sodium leak into the cell making it positive gradually is important. If the funny current increases, like when positive chronotropes are given, the potential reaches towards threshold (red dotted horizontal line) faster and next action potential is generated faster. End result is increase in heart rate – shown in the image as green dotted wave form.
Similarly, if funny current is inhibited (when a negative chronotrope is given), it takes longer time to reach the threshold and next action potential is delayed and the heart rate is slowed. Shown as red dotted wave form.
Once threshold is reached, calcium enters rapidly and an action potential is generated.
Action potentials in Pacemaker cells is due to Calcium influx, not sodium. (Muscle/neuron action potentials are due to Sodium though).
Clinical pearl
Staircase or Treppe effect
The calcium entered from outside into the cardiac muscle cells will be removed by NCX (Sodium calcium exchanger : 3 Sodiums in – 2 Calciums out). When you increase the heart rate, the NCX doesn’t get enough time to pump calcium out and more calcium is available in next heart beat. This additional calcium in each beat is known as Staircase or treppe effect.
Cardiac glycosides
The inhibition of Sodium pump will lead to accumulation of Sodium inside the heart muscle cells. When sodium is already accumulated in the cell, more sodium cannot be pumped by the NCX channel into the cell. If NCX doesn’t work, net effect will be more Calcium inside the cell to improve contractility. This is why we give glycosides like Digitalis in heart failure to improve contractility.
Conduction of Action potential
Saltatory conduction as a product of myelination spaced by nodes of Ranvier
Importance of myelination in terms of speed and energy efficiency
Relation between nerve size and conduction speed (Note: knowledge of the classification of nerve fibres (A, B, I, II etc) is NOT required)
Curriculum
Saltatory conduction
Nerve fibers can be Myelinated or non-myelinated.
Myelin is an insulator and myelinated fibers will have nodes of Ranvier along the nerve which lack myelin. The depolarization jumps from one node to another and is called saltatory conduction.
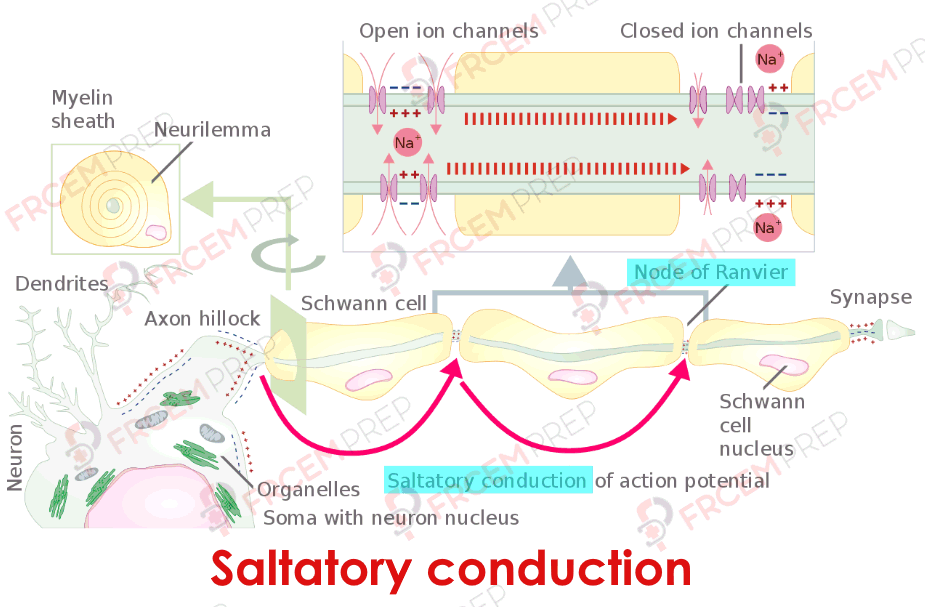
Saltatory conduction is ~50 times faster than in the fastest unmyelinated fibers.
A nerve fiber can transmit impulses in either direction if stimulated in the middle of the axon;
The impulse travels in one direction only.
Fiber diameters and conduction velocities
Smallest diameter unmyelinated fibres – slowest conducting
Largest myelinated fibers – fastest conducting.