Definition
Its importance as the transport mechanism for water based upon gradients.
Iso- hypo- and hyper-tonicity: Differences between each and consequences of ingestion of fluids of each type.
Curriculum
Definition : Osmosis is the passive movement of water across a semi-permeable membrane from regions of low solute concentration to those of higher solute concentration.
Osmotic potential depends on the number of osmotically active particles (molecules) per liter. Biological membranes are semi-permeable. They selectively allow the free movement of water but restrict solutes.
The osmolality of plasma is ∼290 mOsmol/kg H2O.
Tonicity
Tonicity is another way of saying osmolarity in comparison terms.
When some fluid has similar osmolarity to the plasma ( ∼290 mOsmol/kg H2O ), we call it as Isotonic solution.
If a solution has higher osmolarity (concentrated) than plasma, we call it Hypertonic.
If a solution has lower osmolarity (diluted) than plasma, we call if Hypotonic solution.
Consequences of ingestion
Now lets take a look at the consequences of consuming different types of fluids into the body.
Before we move into the discussion, note that any fluid that enters the plasma will freely enter the interstitial space. As a general rule, extracellular compartment has 2 spaces – plasma and interstitial space and fluids/small ions/solutes will freely move between them. Only larger plasma proteins cannot move across the capillaries. Protein content will be different in these 2 spaces but rest of the components could be similar.
When it comes to fluid/solute movement between the cell and extracellular space, as the cell is covered with semipermeable membrane, only water can move from low solute concentration (Hypotonic) space to high solute concentration space (hypertonic) by Osmosis.
When you give any fluid (any tonicity fluid), it gets absorbed into capillaries and plasma volume increases first and concentration changes based on given fluid tonicity.
First things first.
Isotonic
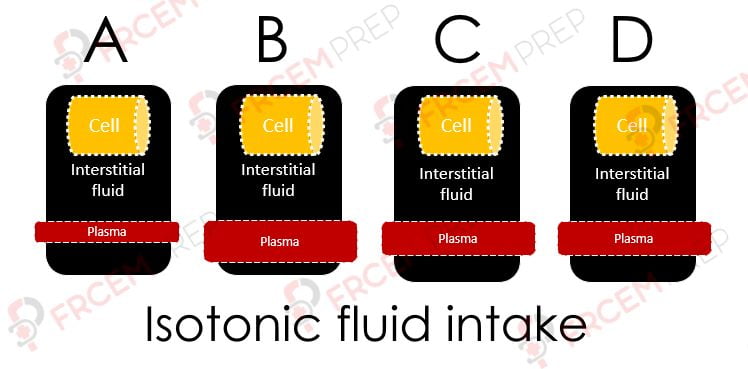
A – If this is the initial state of the fluid spaces, on giving isotonic fluid, the status moves to B
B – Here, the given fluid has entered into Plasma first
C – As capillaries are highly permeable (except for proteins/ large molecules), the same fluid moves into interstitial space.
Note that the osmolarity of both compartments remain the same as given fluid is isotonic, but both volumes will increase..
D – As osmolarity remains the same, there won’t be any movement of fluid between the cell and outside the cell.
Hypotonic
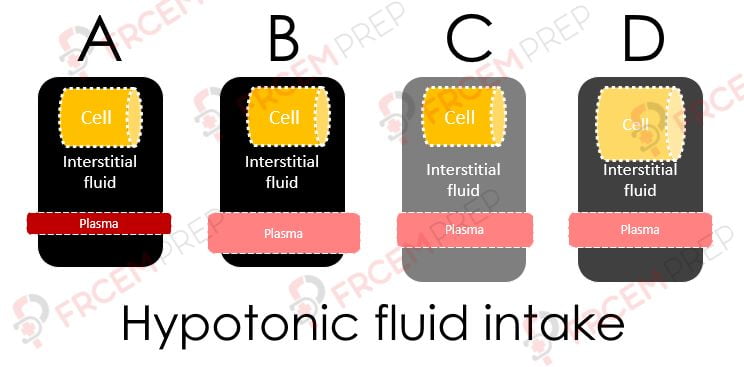
A – If this is the initial state of the fluid spaces, on giving hypotonic fluid, the status moves to B
B – Here, the given fluid has entered into Plasma first and dilutes it (becomes hypotonic)
C – As capillaries are highly permeable (except for proteins/ large molecules), the same fluid moves into interstitial space.
Note that the osmolarity of both compartments decrease and both volumes will increase..
D – As osmolarity of ECF is low, water moves from ECF to ICF and the cell is swollen.
Hypertonic
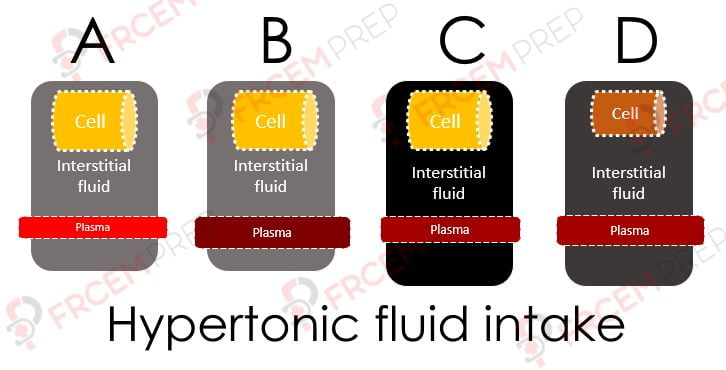
A – If this is the initial state of the fluid spaces, on giving hypertonic fluid, the status moves to B
B – Here, the given fluid has entered into Plasma first and it gets concentrated (darker in pic)
C – As capillaries are highly permeable (except for proteins/ large molecules), the same fluid moves into interstitial space.
Note that the osmolarity of both compartments increases and both volumes will increase..
D – As ECF osmolarity is higher, water will move out of cell and cell shrinks in size.